SPM-TH
Scanning Probe Microscopy Theory & Nanomechanics GroupForces and Transport in Nanostructures
Research Highlights
Molecular identification and bonding information from high-resolution AFM
High-resolution atomic force microscopy (HR-AFM) is able to image the internal structure of molecules, resolves features in the intermolecular regions of weakly bonded molecules, discriminates between bond orders in aromatic compounds, and has opened the door to following or even inducing on-surface chemical reactions. This revolution in molecular imaging has sparked a strong debate about the interpretation of the experiments and the development of new capabilities like chemical identification.
Here, we introduce an efficient method to simulate HR-AFM images with CO probes. Our model explicitly takes into account the charge densities of the sample and the probe for the calculation of the short-range (SR) interaction and retains ab initio accuracy with only two parameters, that are essentially universal, independent of the number of chemical species and the complexity of the bonding topology. The application to molecular images shows a strong dependence on the stoichiometry and bonding configuration that precludes the chemical identification of individual atoms based on local force-distance curves. However, we have identified features in the 2D images and 3D force maps that reflect the highly anisotropic spatial decay of the molecular charge density and provide a way towards molecular identification. The model treats SR and electrostatics interactions on an equal footing and correctly pinpoints the Pauli repulsion as the underlying interaction responsible for the bond order discrimination in C60. Finally, we settle the controversy regarding the origin of the intermolecular features, discarding the effect of the charge redistribution associated with the H bonds, and linking them with the overlap of the wave functions of the atoms that constitute the bond. This overlap creates saddle regions in the potential energy landscape that are sensed by the probe
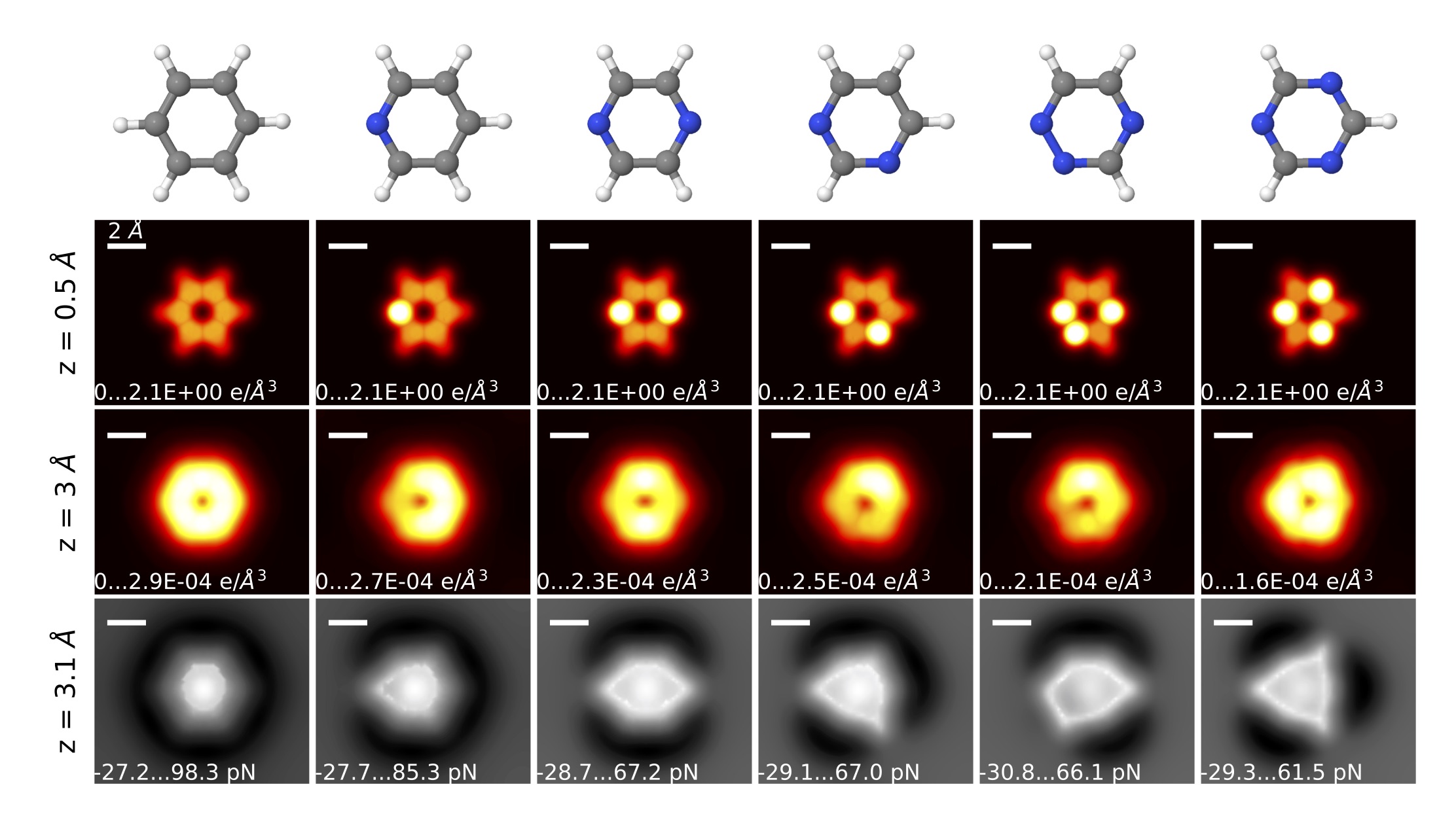
Fig. 1. Six-membered rings with N heteroatoms. From left to right: benzene, pyridine, pyrazine, pyrimidine, 1,2,4-triazine, and an s--triazine. From top to bottom: schematic representation of molecules, charge density at 0.5 and 3 Å from the plane of the molecule, and total force maps (including the effect of CO tilting) at z = 3.1 Å. Although the charge density, and, thus, the N-C repulsive asymmetry seems to be very much dependent on the stoichiometry and bonding configuration, there is a feature that can be associated to the electronic configuration of the N: As in pyridine, the rest of the N heteroatom 6-membered rings' AFM images have sharper vertices around the N atoms as compared to the C-H groups. Far from the plane of the molecule (z=3 Å), the charge density associated with the N lone pair overflows laterally more than the C-H groups. The tip tilt, which emphasizes the saddle lines of the PES, highlights the asymmetry of the charge density around the N atoms. Thus, both the 3D force maps and the sharper vertices disclosing the location of the N atoms in the AFM images provide a fingerprint for the studied molecules.
Michael Ellner, Pablo Pou, and Rubén Pérez
Molecular identifcation, bond order discrimination and apparent intermolecular features in atomic force microscopy studied with a charge density based method
ACS Nano 13, 786--795 (2019).
(DOI link)
(pdf)
(supp. info)